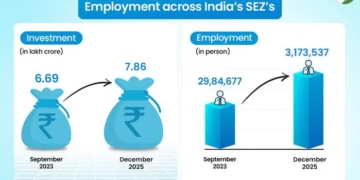
- Agreement allows for a range of potentially best-in-class therapy options as monotherapy and fixed dose combination with Roche’s lead incretin asset CT 388
- Collaboration will complement Roche’s portfolio in the field of cardiovascular, renal, and metabolic (CVRM) diseases
- Obesity is a heterogeneous disease with over 200 related comorbidities, including cardiovascular and metabolic diseases, and is expected to impact over 4 billion people globally by 2035
Basel, 12 March 2025 – Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that it has entered into an exclusive collaboration and licensing agreement with Zealand Pharma (Nasdaq Copenhagen: ZEAL) (CVR-no. 20045078). Under the terms of this agreement, the two companies will collaborate to co-develop and co-commercialise petrelintide, Zealand Pharma’s amylin analog as a standalone therapy as well as a fixed-dose combination with Roche’s lead incretin asset CT-388.
In recent years, scientific advances in the field of incretins and an increased understanding of the disease biology have significantly changed the possibilities to treat obesity and its numerous associated health conditions (comorbidities). With these advances, a large opportunity remains to provide new treatments that offer improved efficacy, safety, quality, and durability of weight loss.
Petrelintide, currently in phase 2 clinical development, is a long-acting amylin analog suitable for once-weekly subcutaneous administration. The available clinical data suggests its potential to become a best-in-class amylin monotherapy, with improved tolerability compared to current weight management treatments and to expand into adjacent indications.
The combination of petrelintide with Roche’s dual GLP-1/GIP receptor agonist CT-388 will further strengthen and expand Roche’s pipeline in the field of cardiovascular, renal, and metabolic (CVRM) diseases. This combination offers the opportunity for best-in-disease efficacy while potentially offering enhanced tolerability.
Teresa Graham, CEO Roche Pharmaceuticals: “We are excited to collaborate with Zealand Pharma and develop this promising therapy, which we hope will provide people living with obesity and related comorbidities a new treatment option. We share the vision to develop petrelintide as a future foundational therapy. By combining petrelintide with our Pharmaceuticals portfolio and with our Diagnostics expertise in cardiovascular and metabolic diseases, we are aiming to transform the standard of care and positively impact patients’ lives.”
Adam Steensberg, President and Chief Executive Officer of Zealand Pharma: “We are thrilled to announce this transformational partnership, aiming to maximise the full value of petrelintide to the benefit of people living with overweight and obesity. With relentless focus on innovation, a global manufacturing network and commercial reach, a complementary portfolio of clinical programmes in obesity, and importantly a shared vision for petrelintide, we consider Roche the ideal partner for Zealand Pharma. We strongly believe that petrelintide holds potential as a foundational therapy for weight management, addressing unmet medical needs among the majority of people living with overweight and obesity, both as stand-alone therapy and in combination with other agents. This collaboration with Roche is a step change to realise this vision, while solidifying Zealand Pharma as a key player in the future management of obesity”.
Terms of the Agreement
This collaboration agreement covers the co-development and co-commercialisation of petrelintide to unlock the full value of the asset. As a part of this agreement, Zealand Pharma and Roche will co-commercialise petrelintide in the U.S. and Europe, whereas Roche will obtain exclusive rights to commercialisation in the rest of the world. Roche will be responsible for commercial manufacturing and supply.
Under the terms of the agreement, Zealand Pharma will receive upfront cash payments of USD 1.65 billion, including USD 1.4 billion due upon closing and USD 250 million over the first two anniversaries of the collaboration. Zealand Pharma is also eligible for development milestones of USD 1.2 billion primarily linked to initiation of Phase 3 trials with petrelintide monotherapy and sales-based milestones of USD 2.4 billion, for a total consideration to Zealand Pharma of up to USD 5.3 billion. Profits and losses for petrelintide and petrelintide/CT-388 will be shared on a 50/50 basis in the U.S. and Europe, and Zealand Pharma is eligible to receive tiered double-digit royalties up to high teens % royalties on net sales in the rest of the world.
Zealand Pharma will pay Roche USD 350 million, offsettable against milestone payments, for the petrelintide/CT-388 fixed-dose combination product or next-generation petrelintide combination products being pursued under the collaboration agreement.
The closing of the transaction is subject to regulatory approvals and other customary closing conditions. The parties expect that the transaction will close in Q2 2025.