“From Domestic Production to International Markets”

Introduction – Linking Public Health with Economic Progress
Health functions both as a driver and an outcome of economic development, and the pharmaceutical industry occupies a unique position at this intersection. By ensuring the availability of essential medicines and vaccines, the industry plays a critical role in advancing public health outcomes and societal welfare, while concurrently generating employment, reinforcing supply-chain resilience, and contributing to broader socio-economic development. Over time, the Indian pharmaceutical industry has emerged as one of the world’s most distinctive and socially significant sectors, positioning India among the largest and most technologically advanced pharmaceutical producers globally.
India in the Global Pharmaceuticals Market
India’s standing as the Pharmacy of the World is rooted in the unique combination of affordable pricing and assured quality, which has made Indian medicines widely preferred across global markets. Cost-efficient manufacturing, supported by a strong scientific workforce and scale, has enabled the consistent supply of essential drugs without compromising standards.
The Indian pharmaceutical industry ranks 3rd globally by volume and 11th by value, with more than 3,000 companies and 10,500 manufacturing units. The domestic pharmaceutical market, valued at USD 60 billion, is projected to reach USD 130 billion by 2030. As per Economic Survey 2025-26, in FY25, the sector’s annual turnover reached Rs. 4.72 lakh crore, with exports growing at a CAGR of 7 per cent over the last decade (FY15 to FY25). India is the largest global supplier of generic medicines, accounting for around 20 per cent of global supply, manufacturing about 60,000 generic brands across 60 therapeutic categories. By expanding access to affordable HIV treatment and emerging as a leading global supplier of cost-effective vaccines, the pharmaceutical industry continues to advance public health outcomes domestically and internationally while generating economic opportunities.

India hosts the highest number of manufacturing plants approved by the United States Food and Drug Administration (USFDA) outside the United States of America, reinforcing international confidence in the safety and quality of Indian pharmaceuticals. There are about 500 active pharmaceutical ingredient (API) manufacturers, accounting for nearly 8 per cent of the global API industry.
Pharmaceutical Exports – Global Reach and Investment Growth
India is the global leader in the supply of Diphtheria, Tetanus, and Pertussis (DPT), Bacillus Calmette-Guerin (BCG), and measles vaccines. Indian manufacturers provide about 60 percent of vaccine supplies to the United Nations International Children’s Emergency Fund (UNICEF), meet 40-70 per cent of global demand for DPT and BCG vaccines, and account for 90 per cent of the World Health Organization’s (WHO) measles vaccine demand. This highlights the robustness of Indian pharmaceutical exports and their substantial integration within global healthcare supply networks.
Export Performance
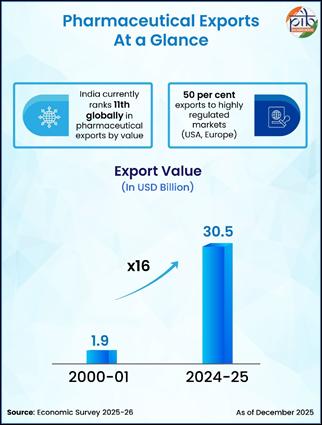
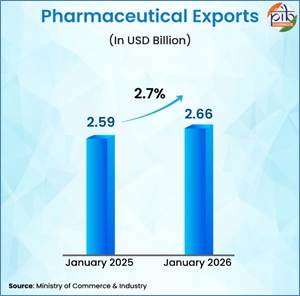
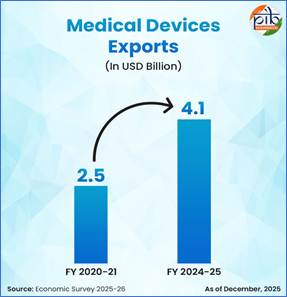
As per the Economic Survey 2025-26, India currently ranks 11th globally in pharmaceutical exports by value, with exports to 191 countries in 2024-25, of which 50 percent are directed to highly regulated markets such as the United States and Europe, reflecting wide international acceptance of Indian medicines. In 2024-25, pharmaceutical exports stood at USD 30.5 billion, a nearly 16-fold increase from USD 1.9 billion in 2000-01. Export momentum has also remained strong every month, with Drugs and Pharmaceuticals exports rising by approximately 2.70 per cent from USD 2.59 billion in January 2025 to USD 2.66 billion in January 2026. Further, medical device exports have grown significantly from USD 2.5 billion in 2020-21 to USD 4.1 billion in 2024-25, with exports to 187 countries in FY25.
India’s pharmaceutical sector has expanded its global presence through rising exports and steady foreign investment, signalling strong international confidence in its manufacturing and regulatory standards. Collectively, these strengths position India as a dependable supplier of quality, affordable medicines and underpin its continued export-driven growth.
Indian pharmaceutical exporters have strategically diversified their export portfolio by expanding shipments to emerging and non-traditional destinations, including Nigeria, Mexico, the United Republic of Tanzania, the Netherlands, France, Brazil, Sri Lanka, Saudi Arabia, and Spain, across bulk drugs, surgical products, and formulations. This targeted market diversification has enhanced export resilience by mitigating exposure to tariff-related risks concentrated in individual markets.
Foreign Direct Investment
The Indian pharmaceutical sector has emerged as a priority destination for foreign investors and is among the top 10 industries attracting foreign investment in India. Foreign investment flows in Drugs and Pharmaceuticals reached 13,193 Crore for the current financial year 2025-26 (up to September). This outlines strengthening investor confidence, supported by expanding production capacity, regulatory stability, and India’s positioning as a global manufacturing hub.
Strengthening India’s Pharmaceutical and Medical Device Exports through Trade Agreements
India’s growing network of trade agreements is expanding global market access for its pharmaceuticals and medical devices sectors. These partnerships are strengthening India’s position as a reliable supplier of affordable medicines and medical technologies worldwide. Recent agreements with Europe and New Zealand are expected to further boost exports, investment, and integration into global healthcare value chains.
India-EU FTA

India and the European Union have concluded negotiations for a Free Trade Agreement that significantly expands opportunities for the Indian pharmaceuticals and medical devices sectors. The agreement provides enhanced market access to the EU, valued at approximately USD 572.3 billion, encompassing pharmaceutical products and medical technologies. Tariff liberalisation is expected to improve the global competitiveness of Indian medical devices, while established manufacturing hubs in Maharashtra, Gujarat, Telangana, Karnataka, and Andhra Pradesh are likely to experience export expansion. The agreement is further anticipated to stimulate skilled employment, strengthen Micro, Small, and Medium Enterprise (MSME) participation, and deepen India’s integration into global healthcare value chains.
India-UK CETA
The India-UK Comprehensive Economic and Trade Agreement (CETA), signed in July 2025, creates new opportunities for India’s pharmaceutical and medical devices sectors. Under the agreement, 56 pharmaceutical tariff lines will receive zero-duty market access, making Indian generic medicines more competitive in the UK, which remains India’s largest pharmaceutical export market in Europe.
The agreement also provides duty-free access for several medical devices, including surgical instruments, diagnostic equipment, ECG machines, and X-ray systems. This is expected to reduce costs for Indian manufacturers and strengthen their competitiveness in the UK market.
India-NZ FTA
The India-New Zealand Free Trade Agreement, concluded in December 2025, expands opportunities for Indian pharmaceutical exports by providing zero-duty access to pharmaceutical products across around 90 tariff lines, where earlier duties ranged up to 5%.
These agreements are therefore expected to deepen healthcare trade and support the growth of India’s pharmaceutical manufacturing ecosystem.
Government-led Intervention Supporting the Pharmaceuticals Sector
At the forefront of the Government’s strategy are the Production Linked Incentive schemes, aimed at strengthening domestic manufacturing, reducing import dependence, and boosting exports across pharmaceuticals, bulk drugs, and medical devices. Complemented by bulk drug and medical device parks, industry support initiatives, and research promotion measures, these schemes are expanding manufacturing capacity, enhancing infrastructure, and reinforcing India’s position as a globally competitive pharmaceutical hub.
Production Linked Incentive (PLI) Schemes
The Production Linked Incentive schemes implemented by the Department of Pharmaceuticals form the cornerstone of efforts to strengthen domestic manufacturing and reduce import dependence across the pharmaceutical sector. These schemes have already resulted in the avoidance of imports worth Rs. 3,591 crore of active pharmaceutical ingredients (APIs), key starting materials (KSMs), and drug intermediates (DIs) under PLI schemes for pharmaceuticals and bulk drugs, thereby contributing to a measurable reduction in import dependency and strengthening India’s manufacturing resilience.
| Active Pharmaceutical Ingredient (API) A substance used in the manufacture of a medicinal product that becomes the active ingredient of the final drug. It provides the pharmacological effect or direct therapeutic action in the diagnosis, treatment or prevention of disease. Drug Intermediate (DI) A material formed during the intermediate stages of synthesis that undergoes further processing before it becomes an active pharmaceutical ingredient. Key Starting Material (KSM) A raw material, intermediate or API used in the production of an API that forms a significant structural part of it. It has defined chemical properties and may be commercially sourced or produced in-house. |
PLI Scheme for Pharmaceuticals
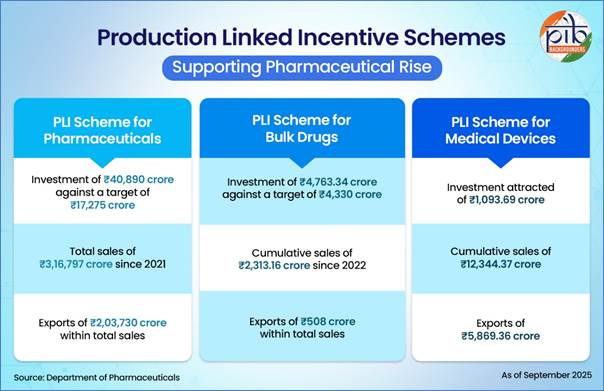
The scheme supports domestic manufacturing of high-value biopharmaceutical products, complex generics, and autoimmune drugs, contributing to significant sales, exports, and employment generation. The investment under the scheme stands at Rs. 40,890 crore against a target of Rs. 17,274.96 crore as of September 2025.
Since its inception in 2021:
- Total sales have reached Rs. 3,16,797 crore up to September 2025.
- Exports account for Rs. 2,03,730 crore within these total sales.
- Approximately 97,000 persons, including contractual and apprentice workers, have been employed under the scheme as of September 2025.
PLI Scheme for Bulk Drugs (APIs/KSMs/DIs)
The scheme aims to ensure a steady supply of critical inputs and reduce reliance on a single source.
As of September 2025:
- Total manufacturing capacity of 55,100 metric tonnes per year has been established for 26 critical APIs/KSMs
- Cumulative sales since inception (2022) have reached Rs. 2,313.16 crore, including exports of Rs. 508.12 crore.
- Investments mobilised under the scheme stand at Rs. 4,763.34 crore against a target of Rs. 4,329.95 crore.
- Employment for 4,929 persons has been generated under the scheme.
PLI Scheme for Promoting Domestic Manufacturing of Medical Devices
This scheme intends to boost domestic manufacturing and attract large investments in the medical devices sector.
As of September 2025:
- The cumulative sales under the scheme stand at Rs. 12,344.37 crore, including exports worth Rs. 5,869.36 crore.
- The scheme has attracted actual investment of Rs. 1,093.69 crore.
Schemes for Infrastructure Development for Bulk Drug and Medical Devices
Scheme for Promotion of Bulk Drug Parks
The Scheme for Promotion of Bulk Drug Parks supports the development of a common infrastructure for bulk drug manufacturing. Under the scheme, three bulk drug parks have been approved and are at various stages of development in the States of Andhra Pradesh, Gujarat, and Himachal Pradesh, through their respective State implementing agencies. As of February 2026, the total project cost of these parks is over Rs.6,306.68 crore, with Central assistance to the tune of Rs.1,000 crore each for creation of common infrastructure facilities (budgetary outlay of Rs.3,000 crore).
Scheme for Promotion of Medical Devices Parks
The scheme aims to provide common testing and laboratory facilities/centres at one place, significantly reducing manufacturing costs and helping create a robust ecosystem for medical device manufacturing in the country. Under this scheme, three parks are being set up and are at an advanced stage of development in Uttar Pradesh (Greater Noida), Madhya Pradesh (Ujjain), and Tamil Nadu (Kanchipuram). The total project cost of these Parks is Rs. 871.11 crore, with a Central grant-in-aid of Rs. 100 crore each for the creation of common infrastructure facilities. As of December 2025, 199 medical device manufacturers have been allotted land across the three parks, total 306.64 acres, and 34 units have commenced construction of their plants.
Other initiatives for Innovation and Improved Access
Research and Innovation
The Scheme for Promotion of Research and Innovation in Pharma MedTech (PRIP) seeks to strengthen India’s pharmaceutical and medical technology ecosystem by moving beyond generic manufacturing towards innovation-driven growth. It supports research, product development, and closer collaboration between industry and academia through two structured components.
- Under Component A, 7 Centres of Excellence have been established, one at each National Institute of Pharmaceutical Education and Research, with an overall outlay of Rs. 700 crore.
- As of November 2025, 111 research projects have been approved, 46 research papers have been published, and 6 patents have been filed under the Centres of Excellence.
- Under Component B, financial assistance is provided to industries, MSMEs, and start-ups for research in identified priority areas in the Pharma MedTech sector.
Access to Affordable Medicines
The Pradhan Mantri Bhartiya Janaushadhi Pariyojana (PMBJP) aims to improve access to affordable, quality generic medicines across the country. Through a wide network of dedicated outlets, the scheme seeks to reduce out-of-pocket expenditure on medicines and ensure that essential drugs and medical devices are available at reasonable prices.
- Under the scheme, as of March 2026, over 18,646 Jan Aushadhi Kendras (JAKs) as of 28.02.2026 are operational.
- The product basket comprises 2,110 medicines, 315 medical devices, and consumables covering 29 therapeutic groups.
- Jan Aushadhi Suvidha Sanitary Napkins are available at Rs. 1 per pad, with over 100 crore pads sold till 31.01.2026. Out of this, over 22.50 crore Jan Aushadhi Suvidha Sanitary Pads have been sold in the FY 2025-26 till 31.01.2026.
- In 2024-25, sales of Rs. 2,022.47 crore resulted in estimated savings of about Rs. 8,000 crore for citizens.
- In 2025-26, up to 30 November 2025, sales of Rs. 1,409.32 crore have led to savings of approximately Rs. 5,637 crore for citizens.
Together, these initiatives demonstrate a coordinated policy approach to strengthening domestic manufacturing, promoting innovation, improving affordability, and enhancing global competitiveness across the pharmaceutical and MedTech sectors.
Biopharma SHAKTI – Strengthening India’s Biopharmaceutical Ecosystem
In the Union Budget 2026-27, the government has proposed the Biopharma SHAKTI (Strategy for Healthcare Advancement through Knowledge, Technology and Innovation) initiative to position India as a global biopharmaceutical manufacturing hub. The initiative will be launched with a total outlay of Rs. 10,000 crore over the next five years and aims to build a strong ecosystem for the domestic production of biologics and biosimilars.

Biopharma SHAKTI will include the creation of a biopharma-focused institutional network, with the establishment of 3 new and the upgradation of 7 existing National Institutes of Pharmaceutical Education and Research (NIPERs). The programme will also support the creation of a network of over 1,000 accredited clinical trial sites across India, strengthening clinical research capacity and supporting innovation in high-value biopharmaceutical therapies.
| Biopharmaceuticals, or biologics, include products such as vaccines, therapeutic proteins, blood components and tissues. They are derived from living sources and are more complex than chemically synthesised small molecule drugs. Biosimilars are follow on versions of approved biologic medicines developed with reference to an originator product. |
Drug Pricing and Regulation – Governance and Compliance
India’s pharmaceutical system is underpinned by a robust regulatory and quality framework that ensures the safety, affordability, and standardisation of medicines across the country.
Central Drugs Standard Control Organisation (CDSCO)
The Central Drugs Standard Control Organisation (CDSCO) serves as India’s national regulatory authority for drugs, medical devices, and cosmetics, overseeing new drug approvals, clinical trial regulation, import and manufacturing licensing, and pharmacovigilance to ensure product safety, quality, and efficacy. Its regulatory mandate is governed by the Drugs and Cosmetics Act, 1940, and associated rules, which are periodically revised to align with evolving scientific and technological advancements.
National Pharmaceutical Pricing Authority (NPPA)
The National Pharmaceutical Pricing Authority (NPPA) is mandated to fix and revise drug prices under the provisions of the Drugs (Prices Control) Order (DPCO), 2013, while monitoring compliance, ensuring the availability of essential medicines, and advising on pharmaceutical policy. The Order is administered by NPPA under powers delegated by the Department of Pharmaceuticals.
Indian Pharmacopoeia Commission (IPC)
The Government of India established the Indian Pharmacopoeia Commission (IPC) to oversee the periodic publication of the Indian Pharmacopoeia, the official compendium of drug standards under the Drugs and Cosmetics Act, 1940. The Pharmacopoeia prescribes standards of identity, purity, and strength for medicines marketed in India, ensuring uniform quality across the pharmaceutical system. Recognised in 19 countries, it serves as a key scientific and regulatory reference, reflecting growing international confidence in India’s technical and regulatory capabilities.
Such a strong regulatory ecosystem is not only safeguarding public health but also reinforcing global confidence in Indian pharmaceuticals. This foundation remains critical to sustaining growth, exports, and innovation in the sector.
Conclusion
India’s pharmaceutical ecosystem has evolved into a globally integrated and policy-supported system that combines scale, affordability, and regulatory credibility. Strong manufacturing capabilities, rising exports, growing foreign investment, and targeted government schemes have collectively strengthened domestic production, reduced import dependence, and expanded global market presence. At the same time, initiatives promoting affordable access, innovation, quality assurance, and regulatory oversight have reinforced public health outcomes and international confidence. The proposed and recently concluded trade agreements with the European Union, the United Kingdom, and New Zealand are expected to further strengthen India’s pharmaceutical and medical devices sector. These agreements will expand market access and deepen India’s global trade linkages in the sector. Together, these elements position India’s pharmaceuticals on a stable, forward-looking trajectory, supporting sustained growth, global engagement, and long-term resilience.






































